Combined Selective Plane Illumination Microscopy
Abstract
Since its initial development in 1976, fluorescence recovery after photobleaching (FRAP) has been one of the most popular tools for studying diffusion and protein dynamics in living cells. Its popularity is derived from the widespread availability of confocal microscopes and the relative ease of the experiment and analysis. FRAP, however, is limited in its ability to resolve spatial heterogeneity. Here, we combine selective plane illumination microscopy (SPIM) and FRAP to create SPIM-FRAP, wherein we use a sheet of light to bleach a 2D plane and subsequently image the recovery of the same image plane. This provides simultaneous quantification of diffusion or protein recovery for every pixel in a given 2D slice, thus moving FRAP measurements beyond these previous limitations. We demonstrate this technique by mapping intranuclear diffusion of NLS-GFP in live MDA-MB-231 cells; SPIM-FRAP proves to be an order of magnitude faster than fluorescence correlation spectroscopy (FCS) based techniques for such measurements. We observe large length-scale (> ~500 nm) heterogeneity in the recovery times of NLS-GFP, which is validated against simulated data sets. 2D maps of recovery times were correlated with fluorescence images of H2B to address conflicting literature on the role of chromatin in diffusion of small molecules. We observed no correlation between histone density and diffusion. We developed a diffusion simulation for our SPIM-FRAP experiments to compare across techniques; our measured diffusion coefficients are on the order of previously reported results, thus validating the quantitative accuracy of SPIM-FRAP relative to well-established methods. With the recent rise of accessibility of SPIM systems, SPIM-FRAP is set to provide a simple and quick means of quantifying the spatial distribution of protein recovery or diffusion in living cells.
Statement of Significance We developed selective plane illumination microscopy combined with fluorescence recovery after photobleaching (SPIM-FRAP) to perform simultaneous FRAP measurements for each pixel in a 2D slice. This technique has the potential to be implemented on almost any light sheet microscope with minimal software development. FRAP studies were previously unable to resolve spatial heterogeneity and FCS techniques require minute-long acquisition times; SPIM-FRAP remedies both of these issues by generating FRAP-based diffusion maps in 4 seconds. This technique can easily be expanded to 3D by photobleaching a single plane and performing light sheet volumetric imaging, which has the benefits of minimal photobleaching and phototoxicity for studying long-term protein turnover. Furthermore, SPIM-FRAP of slowly-recovering structures enables characterization of spatial distortions to measure intracellular stresses.
Read full text here.
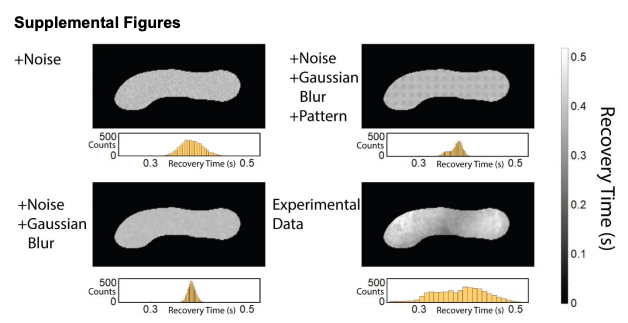
SPIM-FRAP simulated data sets. (Top, Left) Simulation with noise in the recovery curve. Ground truth is a uniform recovery time. (Bottom, Left) Simulation with noise in the recovery curve and Gaussian blurred before analysis. Ground truth is a uniform recovery time. (Top, Right) Simulation with noise in the recovery curve and Gaussian blurred before analysis. Ground truth is a 5x5 pixel array of recovery times that differ by 20%. (Bottom, Right) Experimental data set.

